Using in vivo imaging to understand the innate immune response to infections
White blood cells are our first line of defence against infectious pathogens. Understanding the complex interactions that occur between immune cells and pathogens is important so we can understand how to tackle disease. If we understand these processes then we could develop new drugs to replace antibiotics, that are failing due to the alarming rise in antimicrobial resistant infections.
We use zebrafish to understand these processes. Zebrafish have transparent embryos giving us a window into the whole organism. We use fish with fluorescently labelled white blood cells (macrophages and neutrophils) and infect them with fluorescently labelled Mycobacterium marinum bacteria (fish tuberculosis) to observe the host response to infection down the microscope. This is incredibly informative as we can observe interactions between white blood cell types and bacteria in an intact organism in realtime over the timecourse of early infection.
We are especially interested in hypoxia signalling in white blood cells. Unlike most cells of the body, white blood cells are very efficient in areas of low levels of oxygen (for example at injury sites which lack a blood supply). If we could understand and harness these mechanisms then we may be able to upregulate the immune response to better deal with deadly infections like TB.
We use zebrafish to understand these processes. Zebrafish have transparent embryos giving us a window into the whole organism. We use fish with fluorescently labelled white blood cells (macrophages and neutrophils) and infect them with fluorescently labelled Mycobacterium marinum bacteria (fish tuberculosis) to observe the host response to infection down the microscope. This is incredibly informative as we can observe interactions between white blood cell types and bacteria in an intact organism in realtime over the timecourse of early infection.
We are especially interested in hypoxia signalling in white blood cells. Unlike most cells of the body, white blood cells are very efficient in areas of low levels of oxygen (for example at injury sites which lack a blood supply). If we could understand and harness these mechanisms then we may be able to upregulate the immune response to better deal with deadly infections like TB.
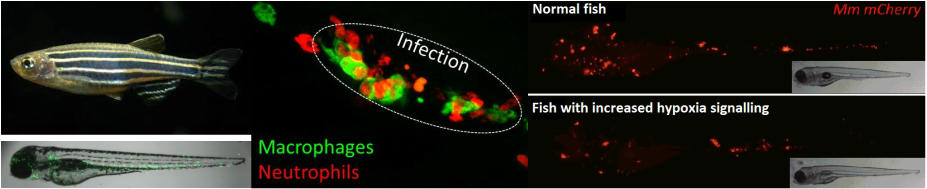
Images from left to right: Adult zebrafish and 3 day old zebrafish larvae with green fluorescently labelled neutrophils (white blood cells). Infection with Mycobacterium marinum leads to immune cell aggregates, or granulomas, involving two immune cell types, macrophages and neutrophils. Red fluorescent labelled bacteria show that the bacterial burden is reduced when hypoxia signalling is increased.
We have shown that increasing hypoxia signalling leads to a lower bacterial burden of zebrafish and aids the host in tackling infections. We are now focused on understanding the dynamic interplay between hypoxia signalling and the response of different populations of white blood cells during infection, with the aim of developing future host-targeted drugs against infectious diseases to replace failing antibiotics.

We work in state-of-the-art laboratories, The Wolfson Laboratories for Zebrafish Models of Infection that opened in 2021 thanks to generous support from The Wolfson Foundation.


